Blood Substitutes
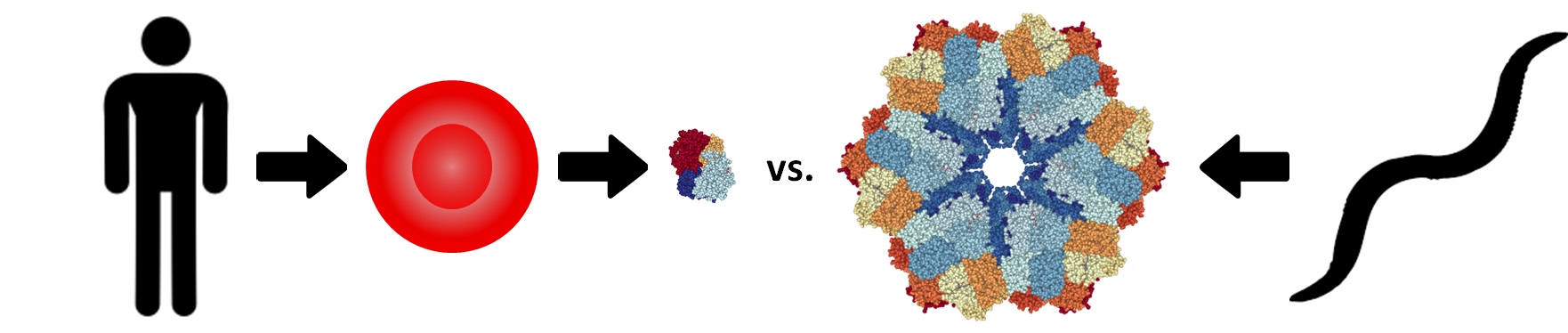
Donated blood is the safest and most effective treatment for patients with severe blood loss. However, there are frequent shortages of rare blood types. In addition, since red blood cells must be constantly refrigerated (never frozen), donated blood is frequently unavailable in remote areas (e.g. battlefields) that lack proper storage facilities. These limitations have motivated the search for a “blood substitute” that can safely and effectively transport oxygen (the most important function of blood), be stored for long periods of time at high temperatures, and should preferably be a universal donor material. Most blood substitutes use human or cow hemoglobin as a starting material, since it is the main component of blood and an excellent oxygen carrier. Unfortunately, those products ultimately failed clinical trials due to a variety of adverse side effects (see below).

The side effects observed previous blood substitutes can be attributed to the fact that human and cow hemoglobin are intracellular proteins that are designed to function within the protective environment of a red blood cell. Therefore, a better approach may be to use one of the naturally extracellular hemoglobins (also known as erythrocruorins) found in most worms and some snails or clams. For example, the erythrocruorin of the common earthworm Lumbricus terrestris (LtEc) has already been shown to have several adaptations (see above) that make it an attractive blood substitute, including: higher structural & thermal stability, slower oxidation rate, and a higher molecular weight (3,600,000 g/mol). In fact, the unusually large size of LtEc allows us to easily purify large amounts of it using just ultrafiltration (500 kDa MWCO TFF). Our preliminary studies show that the purified LtEc can be stored at high temperatures (37-50oC) for several days without any change in structure or function, suggesting that it could be deployed to remote areas. In addition, our preliminary animal studies in both mice and hamsters have shown that LtEc safely delivers oxygen in these animals without causing the adverse side effects observed with human and cow hemoglobin-based blood substitutes.
Grants/Collaborations
With IO Biosciences, expressing LtEc genes in Chinese hamster ovary cells

We sincerely thank the National Institutes of Health (NHLBI) for supporting our research!
NIH NHLBI R15 (AREA):
Publications
Timm B, Abdulmalik O, Chakrabarti A, Elmer J. Purification of Lumbricus terrestris erythrocruorin (LtEc) with anion exchange chromatography. Journal of Chromatography B. 2020 1150:122162.
Timm B, Elmer J. “Enhancing the Stability of Lumbricus terrestris Erythrocruorin (LtEc) for use as a Blood Substitute” in 2017 International Symposium for Blood Substitutes. World Science Publishing/Imperial Press. Accepted, In Press.
Spivack K, Tucker M, Nicholas M, Zimmerman D, Abdulmalik O, Comolli N, Elmer J. Increasing the stability of Lumbricus terrestris Erythrocruorin via Poly(acrylic acid) conjugation. Artificial Cells, Nanomedicine, & Biotech 19:1-8, 2018.
Rajesh A, Zimmerman D, Spivack K, Abdulmalik O, Elmer JJ. Glutaraldehyde cross-linking increases the stability of Lumbricus terrestris erythrocruorin. Biotechnol Prog. 34(2):521-528, 2018.
Zimmerman D, DiIusto M, Dienes J, Abdulmalik O, Elmer JJ. Direct Comparison of Oligochaete Erythrocruorins as Potential Blood Substitutes. Bioengineering & Translational Medicine.
Muzzelo C, Neely C, Shah P, Abdulmalik O, Elmer J. Prolonging the shelf life of Lumbricus terrestris Erythrocruorin for use as a novel blood substitute
Zimmerman D, Dienes J, Abdulmalik O, Elmer J. Purification of diverse hemoglobins by metal salt precipitation.
Roth AD, Elmer J, Harris DR, Huntley J, Palmer AF, Nelson T, Johnson JK, Xue R, Lannutti JJ, Viapiano MS. Hemoglobin regulates the migration of glioma cells along poly(e-caprolactone)-aligned nanofibers.
Elmer J, Palmer AF, Cabrales P. Oxygen delivery during extreme anemia with ultra-pure earthworm hemoglobin. Life Sci. 2012 91:852-859.
Elmer J, Zorc K, Rameez S, Zhou Y, Cabrales P, Palmer AF. Hypervolemic infusion of Lumbricus terrestris erythrocruorin purified by tangential flow filtration. Transfusion. 2012 52:1729-1740.
Elmer J, Palmer AF. Biophysical properties of Lumbricus terrestris Erythrocruorin and its potential use as a red blood cell substitute.
Elmer J, Cabrales P, Wang Q, Zhang N, Palmer AF. Synthesis and biophysical properties of polymerized human serum albumin.
Elmer J, Harris D, Palmer AF. Purification of hemoglobin from red blood cells using tangential flow filtration and immobilized metal ion affinity chromatography.
Elmer J, Buehler PW, Jia Y, Wood F, Harris DR, Alayash AI, Palmer AF. Functional comparison of hemoglobin purified by different methods and their biophysical implications.
Elmer J, Harris DR, Sun G, Palmer AF. Purification of hemoglobin by tangential flow filtration with diafiltration. Biotechnol Prog. 2009 25(5):1402-1410.

